A worker prepares a syringe with the Pfizer COVID-19 vaccine at a clinic at the Reading Area Community College in Reading, Pa.
Matt Rourke / AP Photo

A worker prepares a syringe with the Pfizer COVID-19 vaccine at a clinic at the Reading Area Community College in Reading, Pa.
Matt Rourke / AP Photo

Matt Rourke / AP Photo
A worker prepares a syringe with the Pfizer COVID-19 vaccine at a clinic at the Reading Area Community College in Reading, Pa.
(Washington) — Pfizer and BioNTech are officially asking the Biden administration to authorize the use of their COVID-19 vaccine for children ages 5 to 11.
Pfizer tweeted on Thursday that the companies had submitted their formal request for Emergency Use Authorization of the vaccine to the Food and Drug Administration.
“With new cases in children in the U.S. continuing to be at a high level, this submission is an important step in our ongoing effort against #COVID19,” the pharmaceutical giant said.
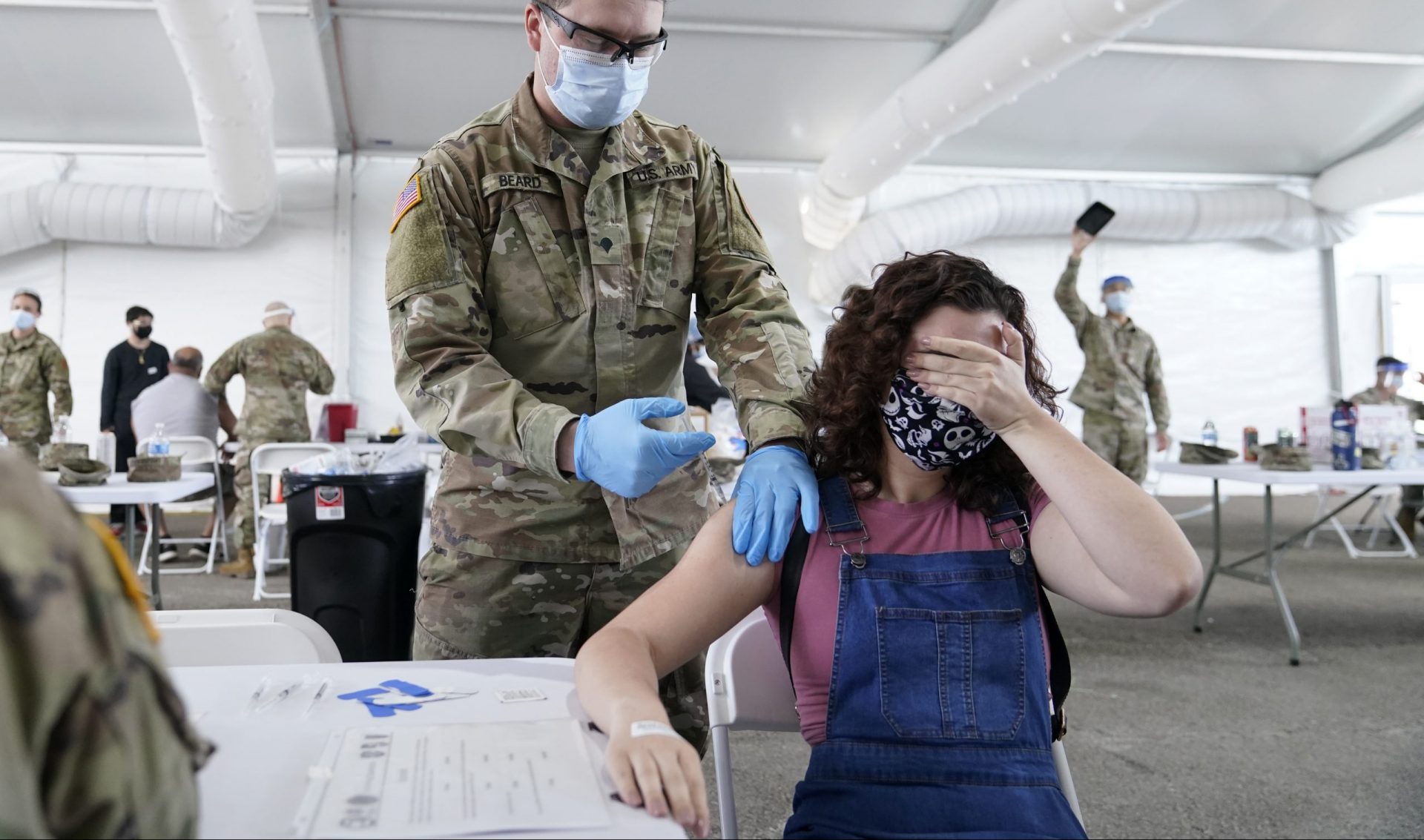
Lynne Sladky / AP Photo
In this April 5, 2021, file photo, Leanne Montenegro, 21, covers her eyes as she doesn’t like the sight of needles, while she receives the Pfizer COVID-19 vaccine at a FEMA vaccination center at Miami Dade College in Miami. Nearly half of new coronavirus infections nationwide are in just five states, including Florida — a situation that puts pressure on the federal government to consider changing how it distributes vaccines by sending more doses to hot spots.
The submission is the latest development in the push to expand use of the vaccine to younger children, a process being closely watched by many anxious parents as the new school year gets underway.
The Pfizer-BioNTech vaccine has full FDA approval for people ages 16 and older, and those between the ages of 12 and 15 can get the shot under the current emergency use authorization.
Last month Pfizer and BioNTech submitted data to the FDA showing that their vaccine produced a “robust” antibody response in kids from 5-11 and had “favorable” safety outcomes.

Sometimes, your mornings are just too busy to catch the news beyond a headline or two. Don’t worry. The Morning Agenda has got your back. Each weekday morning, host Tim Lambert will keep you informed, amused, enlightened and up-to-date on what’s happening in central Pennsylvania and the rest of this great commonwealth.
The days of journalism’s one-way street of simply producing stories for the public have long been over. Now, it’s time to find better ways to interact with you and ensure we meet your high standards of what a credible media organization should be.